Study Guide: Science Term III
- Review the periodic table: groups, periods, energy shells, naturally-occurring elements and synthetic elements.
- Elements in the periodic table: particles in an atom, recognizing what the numbers in the periodic table mean
- Ionic and Covalent Bonds
- Differences and similarities between atoms and molecules, compounds and elements, mixtures and solutions, heterogeneous mixtures and homogeneous mixtures and elements and ions
- Go through the information in your final research because you will be asked about it in the evaluation
- Reflect on the future of chemistry in terms of global water supply, global health and world nutrition
Activity ONE
- Write the title: CHEMISTRY
- Take notes about important information in your science notebook.
- https://youtu.be/vlSOESXQI7o
- Make sure you have these definitions in your science notebook, and understand the differences.
- Matter: anything that occupies space
- Atom: the smallest unit of an element or the smallest unit of anything
- Molecule: two or more atoms bonded together
- Element: substance composed or made up of one type of atom.
- Enter to Campestre's webpage at http://www.campestre.edu.co/estudiantes and enter brainpop.com
- Search for ATOMS
- Watch the video.
- Answer the questions from the Quiz
- In your notebook, do the following activtiy:
- In brainpop, search for SALT.
- Take notes in your science notebook about the important ideas
ACTIVITY TWO
PART I
PART II
PART III
- Write the title IONIC BONDS
- Enter the following page: http://scienceforkids.kidipede.com/chemistry/atoms/ionic.htm
- Read the information carefully, and take notes.
- Answer with as much information as you can in your own words
- What is an ionic bond?
- Draw the example.
PART II
- Write COVALENT BONDS
- Enter the following page: http://scienceforkids.kidipede.com/chemistry/atoms/covalent.htm
- Read the information carefully, and take notes.
- Answer with as much information as you can in your own words
- What is a covalent bond?
- Draw the example.
PART III
- Enter to Campestre's webpage at http://www.campestre.edu.co/estudiantes and enter brainpop.com
- Search for CHEMICAL BONDS
- Watch the video.
- Draw a Venn Diagram in your science notebook
- Compare and contrast ionic bonds and covalent bonds (in your own words)
- After you are done, check with two different students to make sure you have all the possible answers.
ACTIVITY THREE
Part I
Title: THE ATOM
Read about the atom http://www.chem4kids.com/files/atom_intro.html
Write down the most important information in your notebook.
Part II
Title: STRUCTURE OF THE ATOM
Read about the structure of the atom
http://www.chem4kids.com/files/atom_structure.html
Draw a diagram of the atom and explain each part in your own words.
Part III
Title: BONDS
Read about the types of bonds
http://www.chem4kids.com/files/atom_bonds.html
Confirm with the information you already have in your notebook with this information and see if you can add new information about bonds. Draw, summarize, or write the main ideas.
Part IV
Pick different elements on the Periodic Table. Watch the videos. See if you can find similarities among the elements.
Title: THE ATOM
Read about the atom http://www.chem4kids.com/files/atom_intro.html
Write down the most important information in your notebook.
Part II
Title: STRUCTURE OF THE ATOM
Read about the structure of the atom
http://www.chem4kids.com/files/atom_structure.html
Draw a diagram of the atom and explain each part in your own words.
Part III
Title: BONDS
Read about the types of bonds
http://www.chem4kids.com/files/atom_bonds.html
Confirm with the information you already have in your notebook with this information and see if you can add new information about bonds. Draw, summarize, or write the main ideas.
Part IV
Pick different elements on the Periodic Table. Watch the videos. See if you can find similarities among the elements.
WEEK 3
Go through the PowerPoint. Take notes.
| bohrs_model.pptx | |
| File Size: | 2429 kb |
| File Type: | pptx |
Week 4
Take notes in your science notebook about each video.
We will be performing a lab tomorrow. Make sure you understand the difference between solutions and mixtures.
Answer these questions in your notebook.
What is a chemical change? What is a physical change?
What is a chemical change? What is a physical change?
Give examples of chemical reactions.
Give examples of physical reacitons.
If you finish, take notes on the following video.
Give examples of physical reacitons.
If you finish, take notes on the following video.
Review the PowerPoint from Week 3.
TAKE NOTES...
| elements,_mixtures_and_compounds.pptx | |
| File Size: | 500 kb |
| File Type: | pptx |
| properties_of_matter.pptx | |
| File Size: | 541 kb |
| File Type: | pptx |
Cool Information
HOMEWORK WEEK 5 (During "Fiestas Tradicionales")
Homework # 1
In your notebook:
Title: Physical and Chemical changes.
1. Watch the video
2. Take notes
3. Copy the chart in your notebook
4. Write PHYSICAL or CHEMICAL for each example
5.Use a different color to check your answers (double click on the answer box, no cheating, try to the activity before looking at the answers)
http://www.kentchemistry.com/links/Matter/PhysicalChemicalChanges.htm
Homework # 2
In your notebook:
Title: Classify Matter
1. Watch the video
2. Take notes
3. Copy the chart in your notebook
4. Write Element, Compound, Heterogeneous Mixture or Homogeneous Mixture for each example
5.Use a different color to check your answers (double click on the answer box, no cheating, try to the activity before looking at the answers)
http://www.kentchemistry.com/links/Matter/ClassifiyingMatter.htm
In your notebook:
Title: Physical and Chemical changes.
1. Watch the video
2. Take notes
3. Copy the chart in your notebook
4. Write PHYSICAL or CHEMICAL for each example
5.Use a different color to check your answers (double click on the answer box, no cheating, try to the activity before looking at the answers)
http://www.kentchemistry.com/links/Matter/PhysicalChemicalChanges.htm
Homework # 2
In your notebook:
Title: Classify Matter
1. Watch the video
2. Take notes
3. Copy the chart in your notebook
4. Write Element, Compound, Heterogeneous Mixture or Homogeneous Mixture for each example
5.Use a different color to check your answers (double click on the answer box, no cheating, try to the activity before looking at the answers)
http://www.kentchemistry.com/links/Matter/ClassifiyingMatter.htm
In-Class Activity---Week 6 (after Fiestas)
A) Take notes from the video. Then answer the questions.
Video 1: https://www.youtube.com/watch?v=L2Q2q20KaEk
1.How does chemistry affect our lives?
2.Write one idea about the future of chemistry.
B) Takes notes from the video.
Video 2: https://www.youtube.com/watch?v=fPnwBITSmgU
1. In your own words, summarize the important information in the video.
C) Watch the video. Answer the questions below in complete sentences.
Video 3: https://www.youtube.com/watch?v=aVmU3CLxvgU (Water supply)
1. Why is water called the universal solvent?
2. Why being cohesive and adhesive are important properties of water for you?
3. Why do you think high heat capacity of water is mentioned in the video? Why does it seem relevant for the world around us?
4. Explain if the density of water is based on its temperature.
D)Watch the video. Answer the questions below in complete sentences.
Video 4: https://www.youtube.com/watch?v=Y4Kj3cBI4xY (water supply and global health)
1. Why do you think it is important to have clean water?
2. What are the current processes of purification of sea water? Explain.
3. Why aren’t these processes as productive as it is expected?
4. Draw a graph or a diagram to explain the process of switchable water technology.
5. Why is this process positive for the world around us?
E) Watch the video. Answer the questions below in complete sentences.
Video 5: https://www.youtube.com/watch?v=dY18LAHM0ig (World nutrition)
1. Why are Nicolas Appert and Louis Pasteur mentioned in the video?
2. Do you consider that the word “sanitary” should be related to food?
3. What does the Food and Drug Administration do?
4. How do you think chemicals help food to last longer without rotting?
Video 1: https://www.youtube.com/watch?v=L2Q2q20KaEk
1.How does chemistry affect our lives?
2.Write one idea about the future of chemistry.
B) Takes notes from the video.
Video 2: https://www.youtube.com/watch?v=fPnwBITSmgU
1. In your own words, summarize the important information in the video.
C) Watch the video. Answer the questions below in complete sentences.
Video 3: https://www.youtube.com/watch?v=aVmU3CLxvgU (Water supply)
1. Why is water called the universal solvent?
2. Why being cohesive and adhesive are important properties of water for you?
3. Why do you think high heat capacity of water is mentioned in the video? Why does it seem relevant for the world around us?
4. Explain if the density of water is based on its temperature.
D)Watch the video. Answer the questions below in complete sentences.
Video 4: https://www.youtube.com/watch?v=Y4Kj3cBI4xY (water supply and global health)
1. Why do you think it is important to have clean water?
2. What are the current processes of purification of sea water? Explain.
3. Why aren’t these processes as productive as it is expected?
4. Draw a graph or a diagram to explain the process of switchable water technology.
5. Why is this process positive for the world around us?
E) Watch the video. Answer the questions below in complete sentences.
Video 5: https://www.youtube.com/watch?v=dY18LAHM0ig (World nutrition)
1. Why are Nicolas Appert and Louis Pasteur mentioned in the video?
2. Do you consider that the word “sanitary” should be related to food?
3. What does the Food and Drug Administration do?
4. How do you think chemicals help food to last longer without rotting?
Homework
Homework: Bring 3 ideas about how you think chemistry will be used in the future. Use complete sentences and future tense. (ask a parent or family member for ideas)
Week 7 In-Class Activity
- Part I
- In your science notebook, write the title: Future of Chemistry
- Click on the button below. Pick four (4) principles you think are the most important and in your owns words explain each principle in your notebook. (If you click on the blue title, you will have more information to help you)
2. Open Amazing Atom. You will find names of lots of countries and 3 main links about water supply, global health and world nutrition. Click on each of the “I” and read the information on why it is importance of atoms regarding each aspect. Take notes
3. Choose 1 country and take notes regarding each aspect cited above
4. As you are reading this information, think about ways to connect it with 1 or 2 of the 12 principles of green chemistry. Take notes, make sure they are clear. You can use a chart, or a table, but be clear.
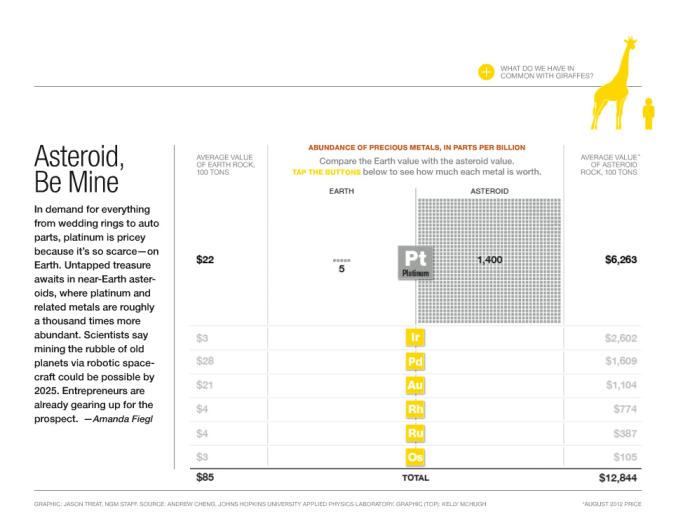
5. Watch this image about Space and Chem elements. Read the information on the left of the graph. Notice that there are some elements taken into account in the graph.
What this meand is that...Asteroids are the most valuable real estate in the Solar System.
Why go to space to mine asteroids? Simple, many of Earth’s precious resources we depend on today originally came from asteroids. Isn’t it about time we went to the source?
Every new frontier that has ever flourished in the history of humanity did so by utilizing the local resources at hand. Space will be no different.
The asteroids we are targeting contain valuable materials such as iron, nickel, rare platinum group metals, and hydrogen and oxygen for rocket fuels. They are energetically much closer to Earth orbit than both the surface of Earth and the Moon. There is no better “local” source of resources to cultivate this frontier. (taken from http://www.planetaryresources.com/#home-asteroids)
Week 8- End of Term Project Instructions
You will research 3 elements. Start working and do a little each week. Organize your time during the next few weeks. You will have time in class to research, but if you know you need more time, make sure to dedicate some time at home. OR make sure to use your time in class wisely.
You will upload the document to your weebly page, so make sure you know where saved (USB, gmail, etc...)
You will upload the document to your weebly page, so make sure you know where saved (USB, gmail, etc...)
| final_project_science_2.docx | |
| File Size: | 13 kb |
| File Type: | docx |
Week 8 -Activities
First part...
Watch the video and follow the instructions below...
https://www.youtube.com/watch?v=QqjcCvzWwww
Write C for Covalent or I for Ionic next to the following compounds.
1. Formed between metal and non-metal atoms ____
2. Formed between non-metal atoms ____
3. Electrons are transferred ____
4. Electrons are shared ____
5. NaCl is this type of compound ____
6. CO2 is an example of this type of compound ____
7. These compounds form crystals ____
8. Weak bonds between molecules but strong bonds between individual atoms exist in these compounds ____
9. There are strong forces between atoms in these compounds ____
10. These molecular compounds have low melting and boiling points and are usually non conductors ____
11. These compounds have a lattice structure ____
12. MgO is an example of this type of compound ____
13. H2 is an example of this type of compound ____
14. Simple molecules or giant structures can form ____
If you’re not sure, check your answers by watching this other video…
https://www.youtube.com/watch?v=KjoQHqgzda8
Part Two
Read this Venn diagram, and copy what you consider is the most important information from it in your notebook
http://creately.com/diagram/example/hkvs18js2/Ionic+Bond+vs.+Covalent+Bond
Part Three
Let's see how much you have learned about the Periodic table symbols
http://www.teachingtreasures.com.au/teaching-tools/science6-10/elements-interactive1-50.htm
4B--- Week 9
What was Pachito trying to teach you?
Explain in your own words using complete sentences and examples from the video.
Do you think covalent bonds will conduct electricity? Why?
Explain in your own words using complete sentences and examples from the video.
Do you think covalent bonds will conduct electricity? Why?
Week 10
Week 10: in class activity
In your notebook copy:
Title: The future of chemistry
Where is chemistry going to and where on Earth (literally) are they going to fins new elements, or even those that we currently need since they are saying that they are all ending!
Watch the following video and answer the questions below: - Video No. 1 : easy water purification secret
http://www.universetoday.com/101552/earth-is-the-most-exotic-place-in-the-universe/
- What material is needed for this process of purification?
- Why is the sun important for this process?
- In which situations would you do this type of purification?
- What is WHO?
Read these texts and watch the video. Answer the question below
Proteins are organic compounds that contain the element nitrogen as well as carbon, hydrogen, and oxygen. Proteins are the most diverse group of biologically important substances and are often considered to be the central compound necessary for life. In fact, the translation from the Greek root word means “first place.” Skin and muscles are composed of proteins; antibodies and enzymes are proteins; some hormones are proteins; and some proteins are involved with digestion, respiration, reproduction, and even normal vision, just to mention a few.
Taken from: http://www.infoplease.com/cig/biology/proteins.html
Lipids are organic compounds that contain the same elements as carbohydrates: carbon, hydrogen, and oxygen. However, the hydrogen-to-oxygen ratio is always greater than 2:1. More important for biological systems, the carbon-to-hydrogen bonds are nonpolar covalent, which means that lipids are fat soluble and will not dissolve in water.
Taken from: http://www.infoplease.com/cig/biology/lipids.html
Carbohydrates consist of the elements carbon (C), hydrogen (H) and oxygen (O) with a ratio of hydrogen twice that of carbon and oxygen. Carbohydrates include sugars, starches, cellulose and many other compounds found in living organisms. In their basic form, carbohydrates are simple sugars or monosaccharides. These simple sugars can combine with each other to form more complex carbohydrates. The combination of two simple sugars is adisaccharide. Carbohydrates consisting of two to ten simple sugars are called oligosaccharides, and those with a larger number are called polysaccharides. Taken from: http://www.scientificpsychic.com/fitness/carbohydrates.html
VIDEO: https://www.youtube.com/watch?v=ClvLhNUz3X8 What Foods the World Eats, Hungry Planet
- Do you consider that you and your family have the best or the worst diet? Why?
- Out of the countries mentioned in the video has the worst diet? Why?
Read the following text and answer the question below:
Contributions made by chemistry
The contributions of chemistry to society are vast and almost numberless. An in-exhaustive list would include
An understanding of chemistry was also key to the development of modern food health and hygiene practices. By understanding the actions of microbes on the human body, these practices have helped eliminate other dread diseases in the Western world, such as dysentery and cholera, as well as many types of food poisoning.
Similarly, to Jenner's vaccines, understanding the chemistry of modern diseases is the key to the ongoing battle against conditions like cancer, AIDS and the common cold. By understanding the chemistry of how, say, cancer cells do what they do inside a human body, we have been able to devise treatments that can in some cases kill the disease, or at least provide an effective treatment regime. Diagnostic tools like dyes,that show up only certain times of tissue on ultrasound readers are also the result of chemical marking processes.
Chemistry was at the heart of understanding, that a chemical sludge pumped from underground had combustible properties that could drive an engine - so without an appreciation of chemistry, your car wouldn't start. Ironically, chemistry is also now at the forefront, of the search for alternative ways to drive car engines without leaving the ecologically harmful residues that current engines do.
From oil comes plastics - but only if you understand how to make them, which means, altering the chemical structure of the oil and processing it to synthesize the plastic out of which half of our modern world appears to be made. Likewise, synthetic fibers are called synthetic because they've been developed by chemists to behave in certain ways - without chemistry, there would be no lycra.
Also, in the modern age, as we have become more fastidious about cleanliness and our appearance, we have developed huge industries dedicated to the development of cosmetics and cleansers - all of which are mixtures of chemicals (either naturally or synthetically produced), that have been blended to give us the effects we want without causing us any harm. Likewise, our quest for cleanliness has led us to develop surface cleaners, which are mixtures of entirely different chemicals that will destroy bacteria - and again, our knowledge of chemistry has both advised us of the threat, and helped us master it.
This list is by no means complete - chemistry has had a part in practically every facet of the modern world - but without chemistry, it should be clear that the world we live in, would look very different.
- Which of the contributions named in the text do you consider are the most important? Explain your answer.
In your notebook copy:
Title: The future of chemistry
Where is chemistry going to and where on Earth (literally) are they going to fins new elements, or even those that we currently need since they are saying that they are all ending!
Watch the following video and answer the questions below: - Video No. 1 : easy water purification secret
http://www.universetoday.com/101552/earth-is-the-most-exotic-place-in-the-universe/
- What material is needed for this process of purification?
- Why is the sun important for this process?
- In which situations would you do this type of purification?
- What is WHO?
Read these texts and watch the video. Answer the question below
Proteins are organic compounds that contain the element nitrogen as well as carbon, hydrogen, and oxygen. Proteins are the most diverse group of biologically important substances and are often considered to be the central compound necessary for life. In fact, the translation from the Greek root word means “first place.” Skin and muscles are composed of proteins; antibodies and enzymes are proteins; some hormones are proteins; and some proteins are involved with digestion, respiration, reproduction, and even normal vision, just to mention a few.
Taken from: http://www.infoplease.com/cig/biology/proteins.html
Lipids are organic compounds that contain the same elements as carbohydrates: carbon, hydrogen, and oxygen. However, the hydrogen-to-oxygen ratio is always greater than 2:1. More important for biological systems, the carbon-to-hydrogen bonds are nonpolar covalent, which means that lipids are fat soluble and will not dissolve in water.
Taken from: http://www.infoplease.com/cig/biology/lipids.html
Carbohydrates consist of the elements carbon (C), hydrogen (H) and oxygen (O) with a ratio of hydrogen twice that of carbon and oxygen. Carbohydrates include sugars, starches, cellulose and many other compounds found in living organisms. In their basic form, carbohydrates are simple sugars or monosaccharides. These simple sugars can combine with each other to form more complex carbohydrates. The combination of two simple sugars is adisaccharide. Carbohydrates consisting of two to ten simple sugars are called oligosaccharides, and those with a larger number are called polysaccharides. Taken from: http://www.scientificpsychic.com/fitness/carbohydrates.html
VIDEO: https://www.youtube.com/watch?v=ClvLhNUz3X8 What Foods the World Eats, Hungry Planet
- Do you consider that you and your family have the best or the worst diet? Why?
- Out of the countries mentioned in the video has the worst diet? Why?
Read the following text and answer the question below:
Contributions made by chemistry
The contributions of chemistry to society are vast and almost numberless. An in-exhaustive list would include
- Vaccines
- Food safety practices
- Treatment programs for diseases
- Diagnostic tools in healthcare
- Plastics
- Synthetic fibers
- An understanding of oil
- Cosmetics and cleaners.
An understanding of chemistry was also key to the development of modern food health and hygiene practices. By understanding the actions of microbes on the human body, these practices have helped eliminate other dread diseases in the Western world, such as dysentery and cholera, as well as many types of food poisoning.
Similarly, to Jenner's vaccines, understanding the chemistry of modern diseases is the key to the ongoing battle against conditions like cancer, AIDS and the common cold. By understanding the chemistry of how, say, cancer cells do what they do inside a human body, we have been able to devise treatments that can in some cases kill the disease, or at least provide an effective treatment regime. Diagnostic tools like dyes,that show up only certain times of tissue on ultrasound readers are also the result of chemical marking processes.
Chemistry was at the heart of understanding, that a chemical sludge pumped from underground had combustible properties that could drive an engine - so without an appreciation of chemistry, your car wouldn't start. Ironically, chemistry is also now at the forefront, of the search for alternative ways to drive car engines without leaving the ecologically harmful residues that current engines do.
From oil comes plastics - but only if you understand how to make them, which means, altering the chemical structure of the oil and processing it to synthesize the plastic out of which half of our modern world appears to be made. Likewise, synthetic fibers are called synthetic because they've been developed by chemists to behave in certain ways - without chemistry, there would be no lycra.
Also, in the modern age, as we have become more fastidious about cleanliness and our appearance, we have developed huge industries dedicated to the development of cosmetics and cleansers - all of which are mixtures of chemicals (either naturally or synthetically produced), that have been blended to give us the effects we want without causing us any harm. Likewise, our quest for cleanliness has led us to develop surface cleaners, which are mixtures of entirely different chemicals that will destroy bacteria - and again, our knowledge of chemistry has both advised us of the threat, and helped us master it.
This list is by no means complete - chemistry has had a part in practically every facet of the modern world - but without chemistry, it should be clear that the world we live in, would look very different.
- Which of the contributions named in the text do you consider are the most important? Explain your answer.
Week 10 (After ice cream reading)
Title: Ice cream – Ice cream is one of people’s favorite desserts around the world, but what is in it?
http://www.acs.org/content/acs/en/education/whatischemistry/adventures-in-chemistry/secret-science-stuff/ice-cream.html Take notes about interesting information.
COPY THE QUESTIONS IN YOUR NOTEBOOK
- What makes ice cream so smooth and fluffy?
- Which ingredient has no taste?
- Did you know it takes more than a freezer to make ice cream cold?
Continue working on your research about your three elements. Here are some helpful websites and resources. (Copy and paste)
http://theodoregray.com/periodictable/
https://www.youtube.com/watch?v=xd4-Uy2FLWc
http://www.nature.com/nchem/journal/v1/n1/full/nchem.139.html (and More Articles like this –which is a the bottom of the page)
http://www.universetoday.com/101552/earth-is-the-most-exotic-place-in-the-universe/
http://www.acs.org/content/acs/en/education/whatischemistry/adventures-in-chemistry/secret-science-stuff/ice-cream.html Take notes about interesting information.
COPY THE QUESTIONS IN YOUR NOTEBOOK
- What makes ice cream so smooth and fluffy?
- Which ingredient has no taste?
- Did you know it takes more than a freezer to make ice cream cold?
Continue working on your research about your three elements. Here are some helpful websites and resources. (Copy and paste)
http://theodoregray.com/periodictable/
https://www.youtube.com/watch?v=xd4-Uy2FLWc
http://www.nature.com/nchem/journal/v1/n1/full/nchem.139.html (and More Articles like this –which is a the bottom of the page)
http://www.universetoday.com/101552/earth-is-the-most-exotic-place-in-the-universe/